A systematic review and thematic synthesis of the barriers and facilitators to physical activity for women after gestational diabetes: a socio-ecological approach
ELYSA IOANNOU,1 HELEN HUMPHREYS,2 CATHERINE HOMER,1 ALISON PURVIS1
1 Sport and Physical Activity Research Centre, Sheffield Hallam University, Sheffield, UK.
2 Centre for Behavioural Science and Applied Psychology (CeBSAP), Sheffield Hallam University, Sheffield, UK.
Address for correspondence: Ms Elysa Ioannou
Collegiate Hall, Sheffield Hallam University, Collegiate Crescent, Sheffield, S10 2NA, UK.
E-mail: E.ioannou@shu.ac.uk
https://doi.org/10.15277/bjd.2023.413
Abstract
Physical activity can reduce risk of type 2 diabetes (T2DM) after gestational diabetes. Understanding barriers and facilitators to physical activity, using a socio-ecological approach, could better direct multi-level interventions. The present review aimed to synthesise barriers and facilitators to physical activity, and to develop an understanding of where, across the socio-ecological model, these factors exist and/or are interrelated. Eligible studies included women with a history of gestational diabetes and a discussion around physical activity. A systematic search of MEDLINE, the Cochrane Library, Web of Science, CINAHL Complete and Scopus was conducted in October 2022. Barriers and facilitators to physical activity were thematically analysed and themes organised according to the socio-ecological model. Twenty-nine studies were included.
Barriers pertained to leisure time physical activity, while other types of activity including housework and transport were overlooked, despite being routine. Partner and family support were vital for engagement with activity, whether emotional support or provision of childcare. Most barriers and facilitators at the social and organisational levels were interrelated with those at the individual level. These findings suggest that multi-level physical activity interventions after gestational diabetes could be most effective.
Br J Diabetes 2023;23:2-13
Key words: physical activity, gestational diabetes, socio-ecological model, type 2 diabetes, barriers, facilitators, women’s health, maternal health
Introduction
Gestational Diabetes Mellitus (GDM) occurs during pregnancy, and its prevalence has been steadily increasing, with the International Diabetes Federation (IDF) reporting a prevalence of 20.6% in the UK in 2021.1 A GDM diagnosis increases the risk of several long- term complications, including increasing the risk of subsequent type 2 diabetes mellitus (T2DM) ten-fold.2,3 Preventing T2DM after GDM is a clinical priority.4
Lifestyle changes, including diet and physical activity (PA), can reduce risk of T2DM by up to 50%.5-7 This level of risk reduction can also be achieved after GDM.8,9 The National Institute for Health and Care Excellence (NICE) recommends promoting healthy lifestyle behaviours after a case of GDM.8 In the UK women with previous GDM can access the “Healthier You” National Diabetes Prevention Program. However, this program was designed for the general population, who may not face the unique barriers present for women with young families, such as other family commitments, lack of childcare and other responsibilities.9,10 This could in part explain why people who do engage with these lifestyle programs tend to be over the age of 65 years,11 and why GDM patients’ participation in prevention interventions is variable.12 Overcoming engagement barriers to lifestyle changes in this population is important for lasting behaviour change and subsequent T2DM risk management.
The barriers to participation and engagement with PA after GDM may not be entirely within an individual’s power to control. The Socio-Ecological Model (SEM) can be used to aid understanding of interrelationships between individuals and the factors associated with their surrounding environments, such as social, physical and policy factors.13 Viewing barriers and facilitators to PA with an SEM lens could therefore improve understanding of the cultural, social and other contextual factors that impact PA for women after GDM.14,15 Peng et al. used the SEM to explore barriers and facilitators to PA for young adult women and highlighted the wider socio-cultural influences on PA and the need for including multi- level strategies to target women’s PA.16 For example, at wider levels, family support was ‘crucial’ to engaging with PA, while family commitments were the most significant barrier to PA for young adult women. It is therefore important to explore whether there are any differences or similarities in the wider barriers and facilitators to PA for women after GDM, to better tailor multi-level strategies aiming to improve PA after GDM.
The only, review to look at barriers and facilitators to lifestyle changes postpartum was published in 2019 by Dennison et al.10 The barriers and facilitators to PA may differ in comparison to those of other lifestyle changes such as diet, since PA may be considered less important and time constraints may limit PA more.17,18 Buelo et al. explored PA-specific barriers and facilitators as part of a mixed methods review, where the qualitative component organised themes according to Dahlgren and Whitehead’s determinants of health model.19 However, the Dahlgren-Whitehead model was designed to explore impacts on health, while the SEM highlights the interrelated systems surrounding and influencing individual behaviour, and therefore provides the structure for a deeper dive into the wider contexts affecting PA. The present review aimed to update these reviews, using a socio-ecological lens, to explore the barriers and facilitators to PA for women after GDM.
Methods
Five databases (MEDLINE, CINAHL, Scopus, Web of Science and Cochrane) and reference lists were searched in October 2022. Three main search themes (combined with ‘AND’) were constructed with the phenomenon of interest (physical activity and T2DM prevention as two separate themes) and sample (women with a history of GDM).20 Within these themes, Mesh and search terms were combined with ‘OR’. Terms were developed from other reviews of barriers and facilitators,18 and lifestyle interventions after GDM.19,21-24
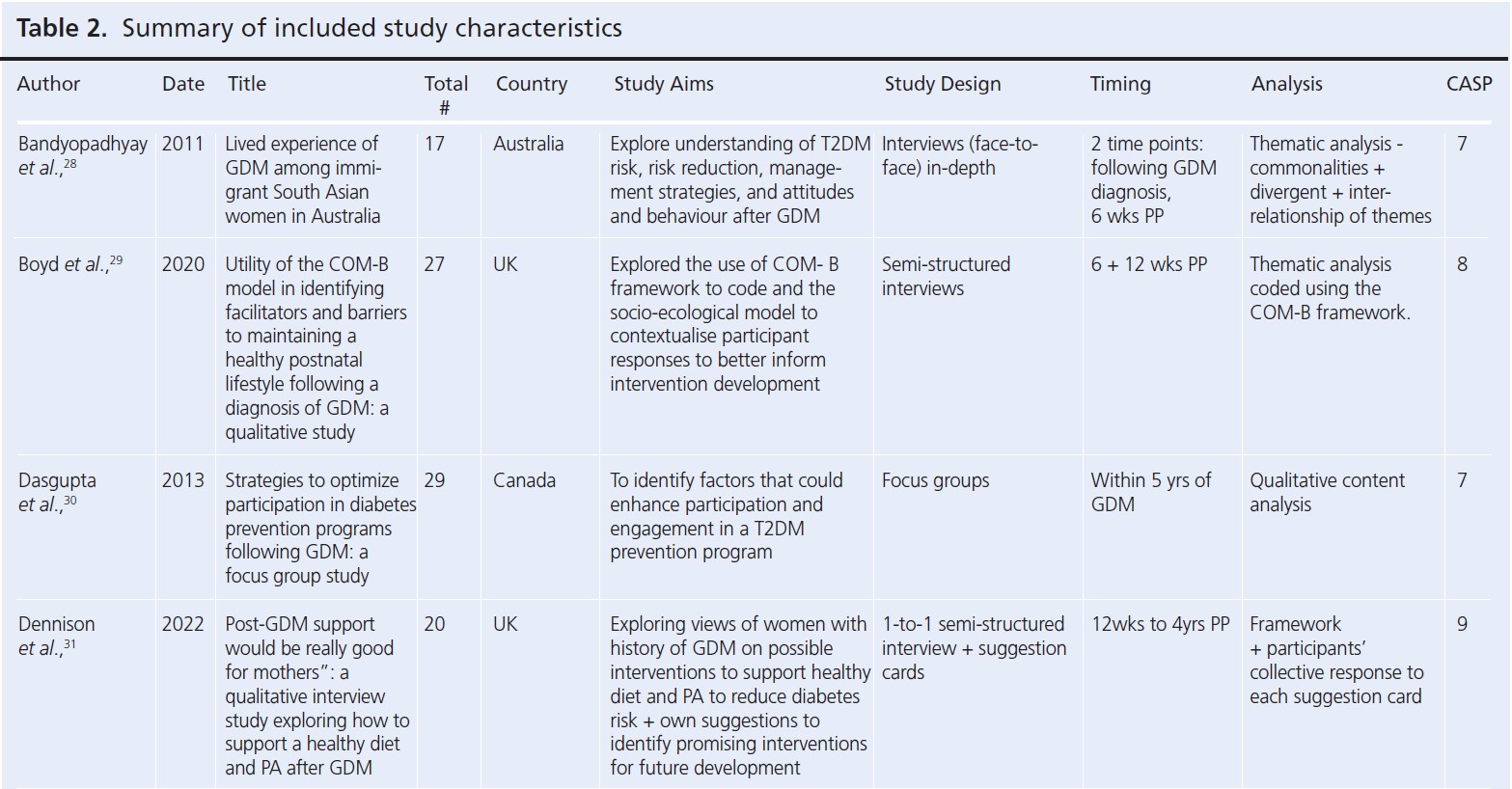
Table 1 summarises the inclusion criteria. The SPIDER tool was used to determine eligibility.25 While studies did not exclusively explore PA postpartum, PA discussions had to be reported in the results, either as part of a lifestyle intervention or general attitudes for lifestyle changes. Title and abstracts were screened by the first author (EI), with a second round of screening at full-text level. A second reviewer (HH) independently processed a random 10% sample of papers at each stage. Disagreements were resolved by discussion. EI used the Critical Appraisal Skills Programmes (CASP) checklist for qualitative research as a quality assessment tool for the studies included in the present review, with a sample discussed with a second reviewer (HH).26
A reflexive thematic analysis was employed, where multiple coders aided reflexivity in interpretations and sense-making from themes.27 Open coding was used inductively, and data were extracted as reported results or participant quotes. Descriptive themes were then organised according to the SEM.14 Themes were grouped into respective levels depending on where they were actionable. This helped view barriers and facilitators through the lens of wider contexts and their influences on individual behaviour,13 and enabled identification of relationships between themes (inter- relationships) i.e. where themes appeared to act across more than one level. Nvivo 12 was utilised by the research team to aid the process of thematic analysis, as the team were all familiar with the software and were able to share the files so all authors could access and review the data and coding.
Results
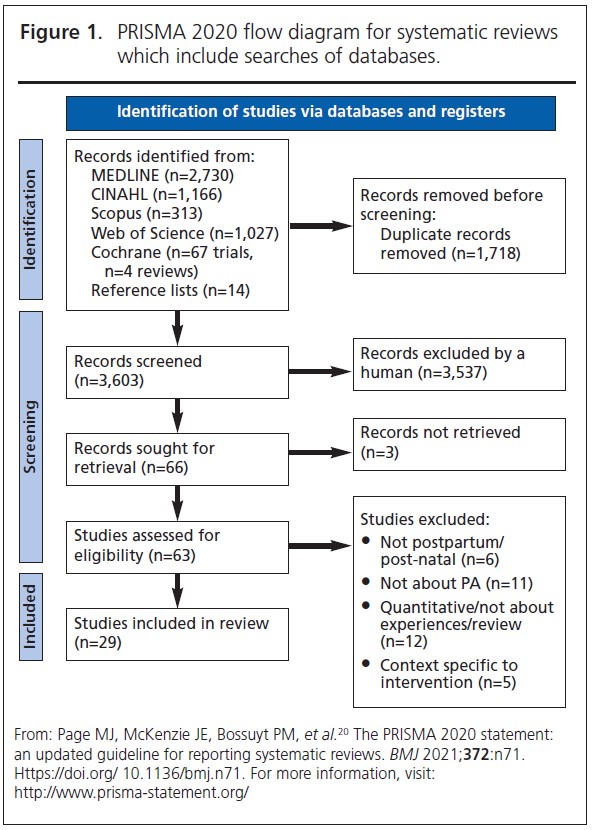
Twenty-nine studies were included (Figure 1).20 At title and abstract stage, 3,603 records were screened and 63 progressed to full-text screening. Articles were excluded if participants were pregnant (n=6), if the studies did not include PA (n=11), and if they were quantitative or review papers (n=12). Summary of included participant characteristics can be seen in Appendix 1.
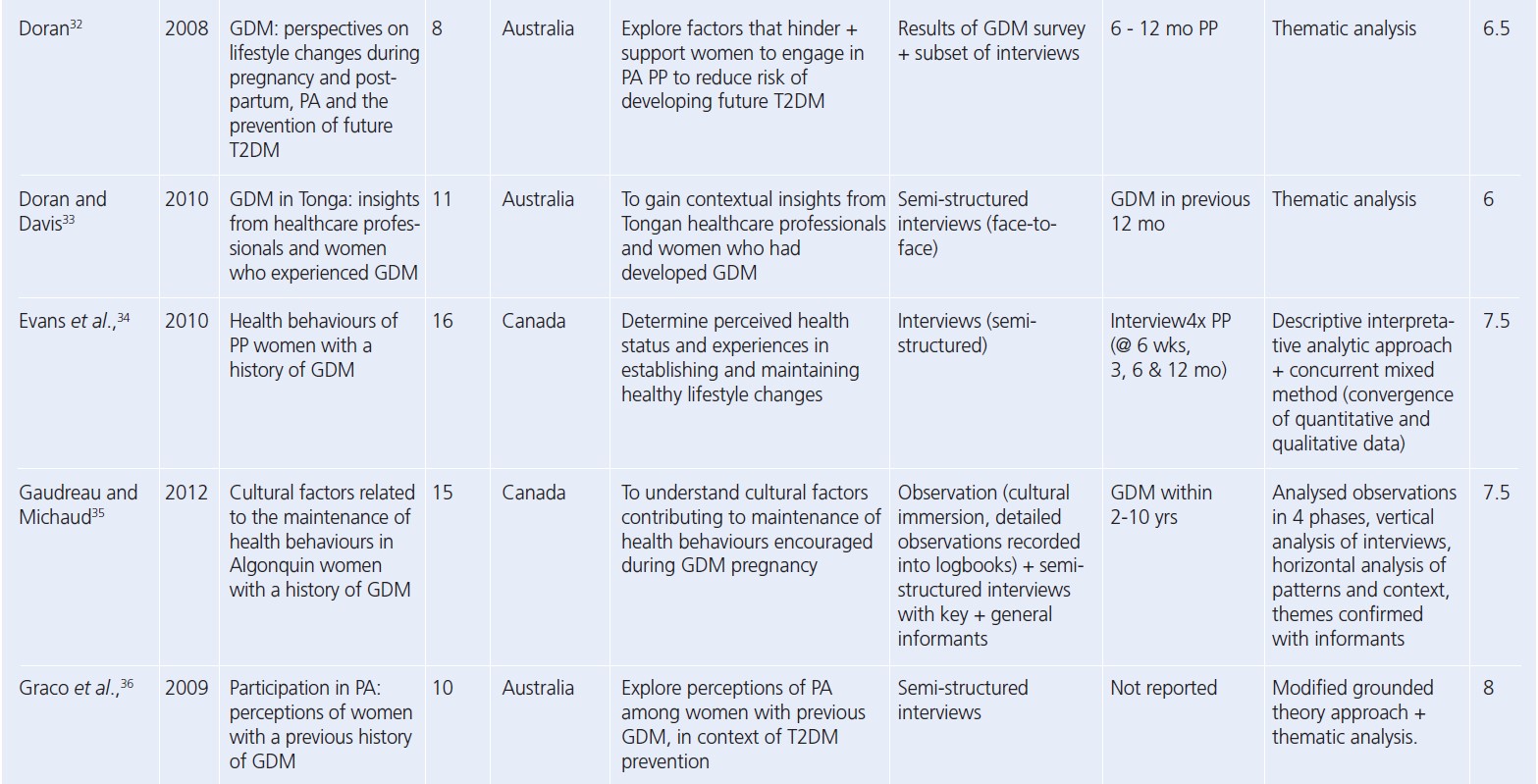
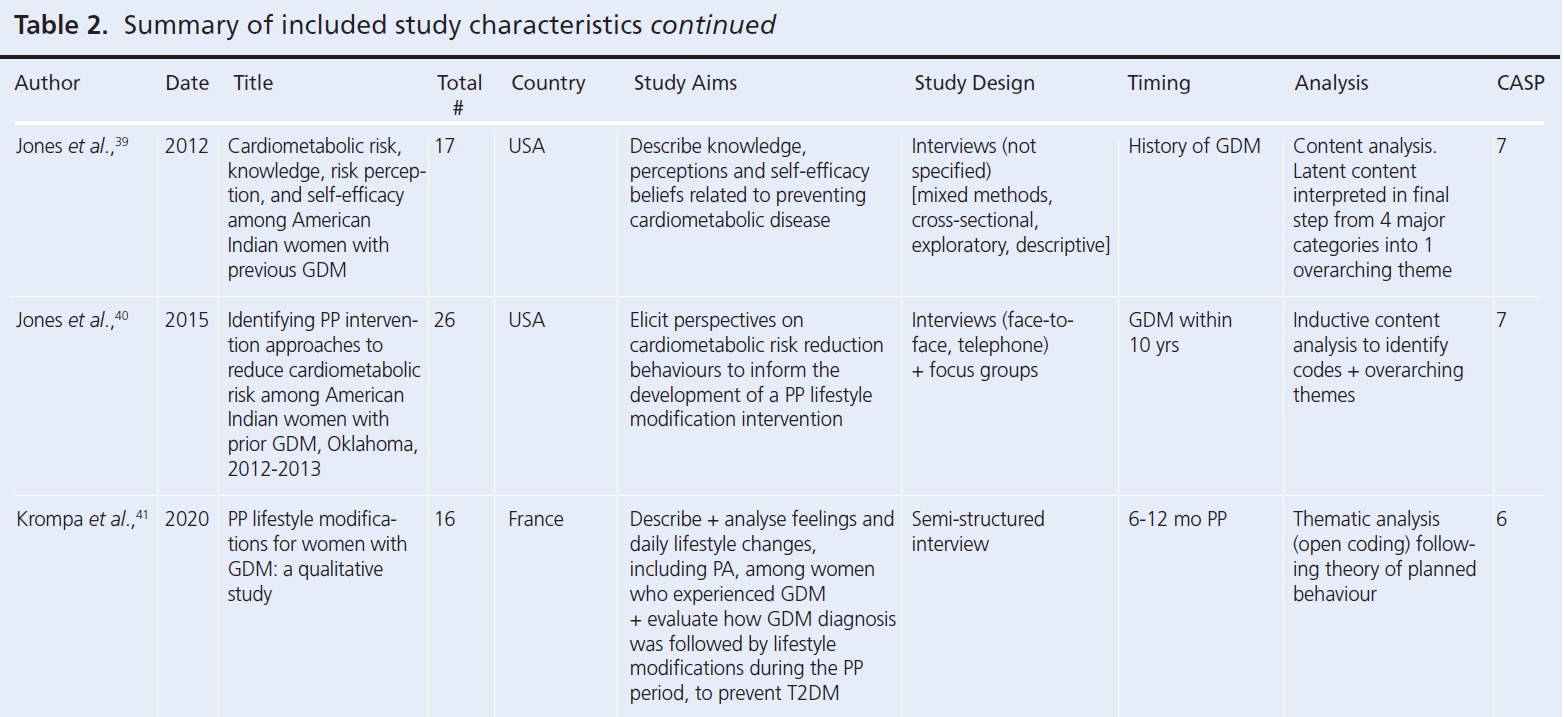
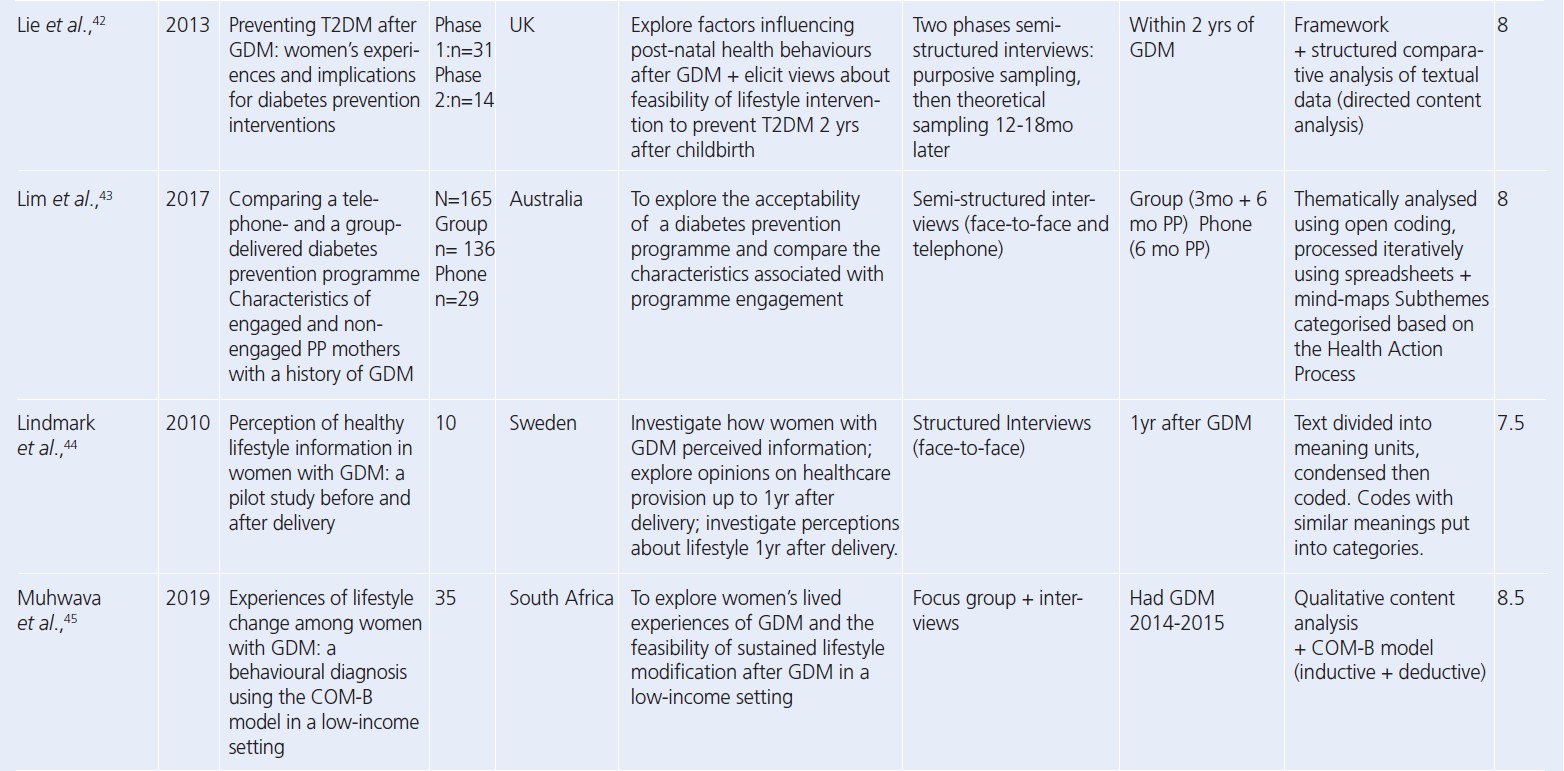
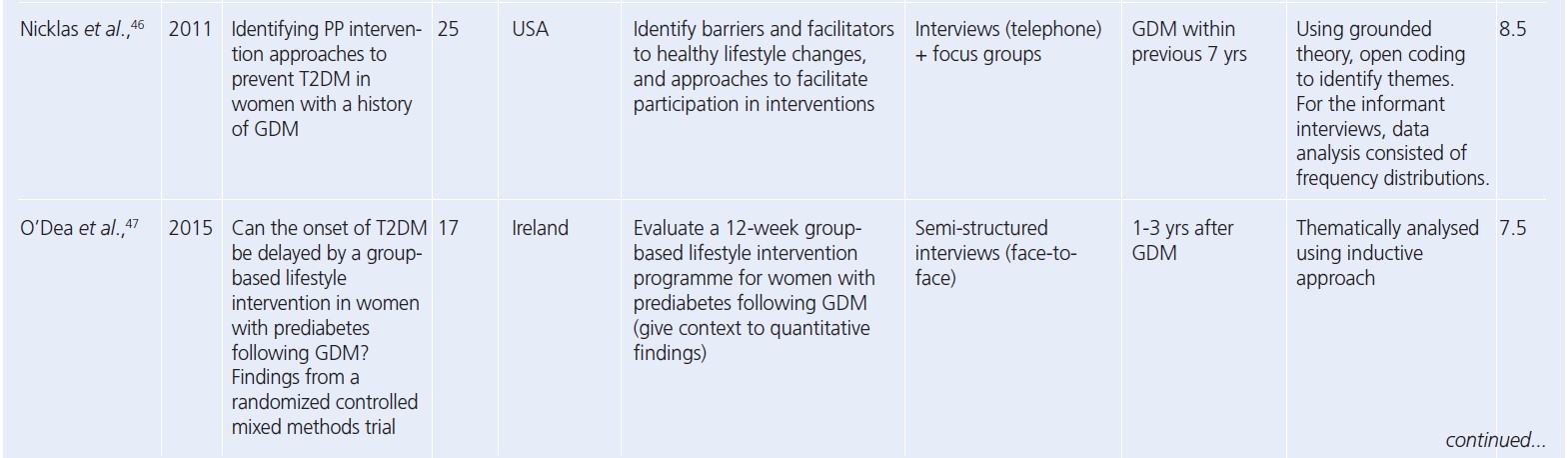
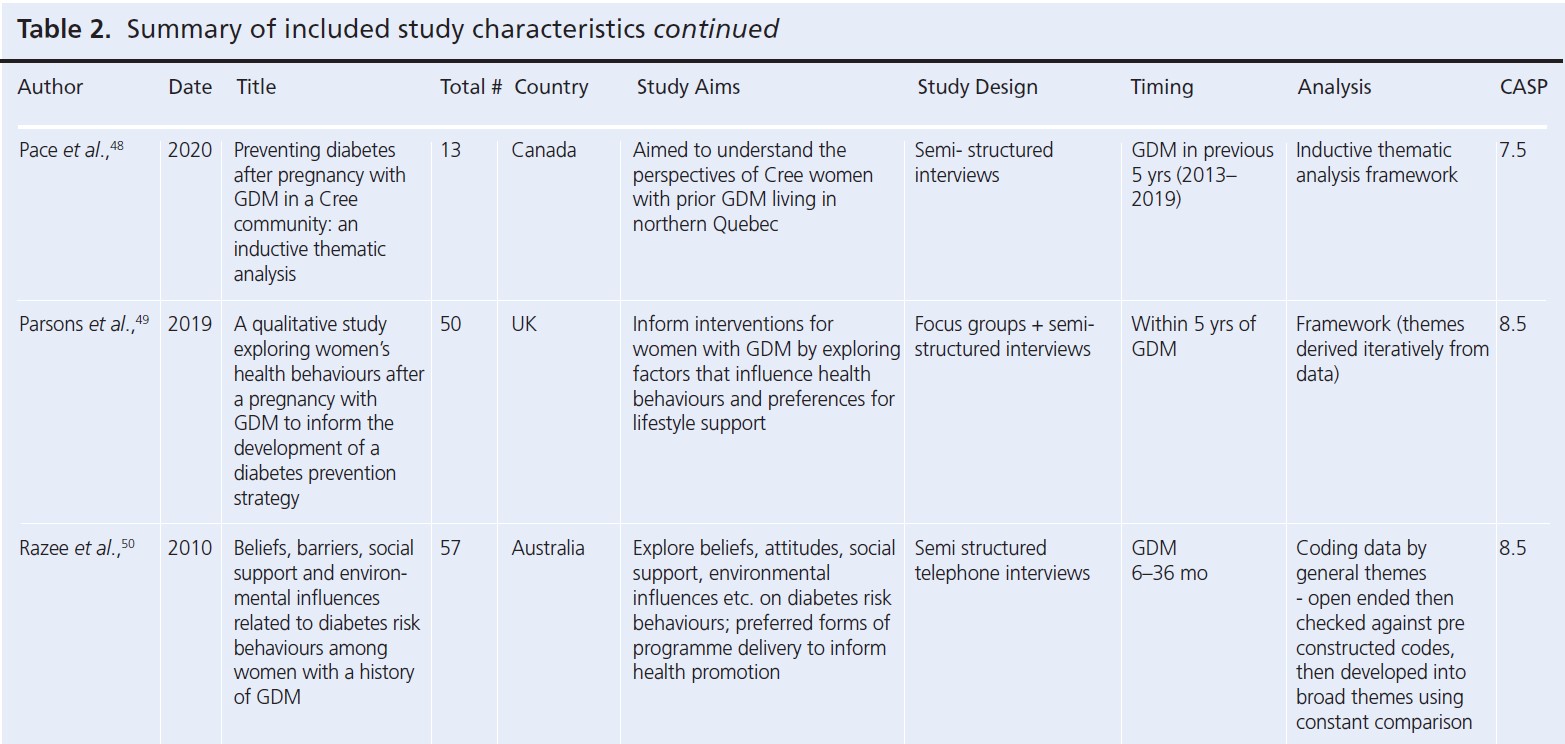
Since the reviews published by Dennison et al. and Buelo et al. in 2019,10,19 nine new published papers were identified. A summary of the study characteristics of all the papers considered in this review is presented in Table 2.28-56 All but four studies had a CASP study quality rating greater than or including seven (n=25). The lowest quality rating of six was given to two separate studies.
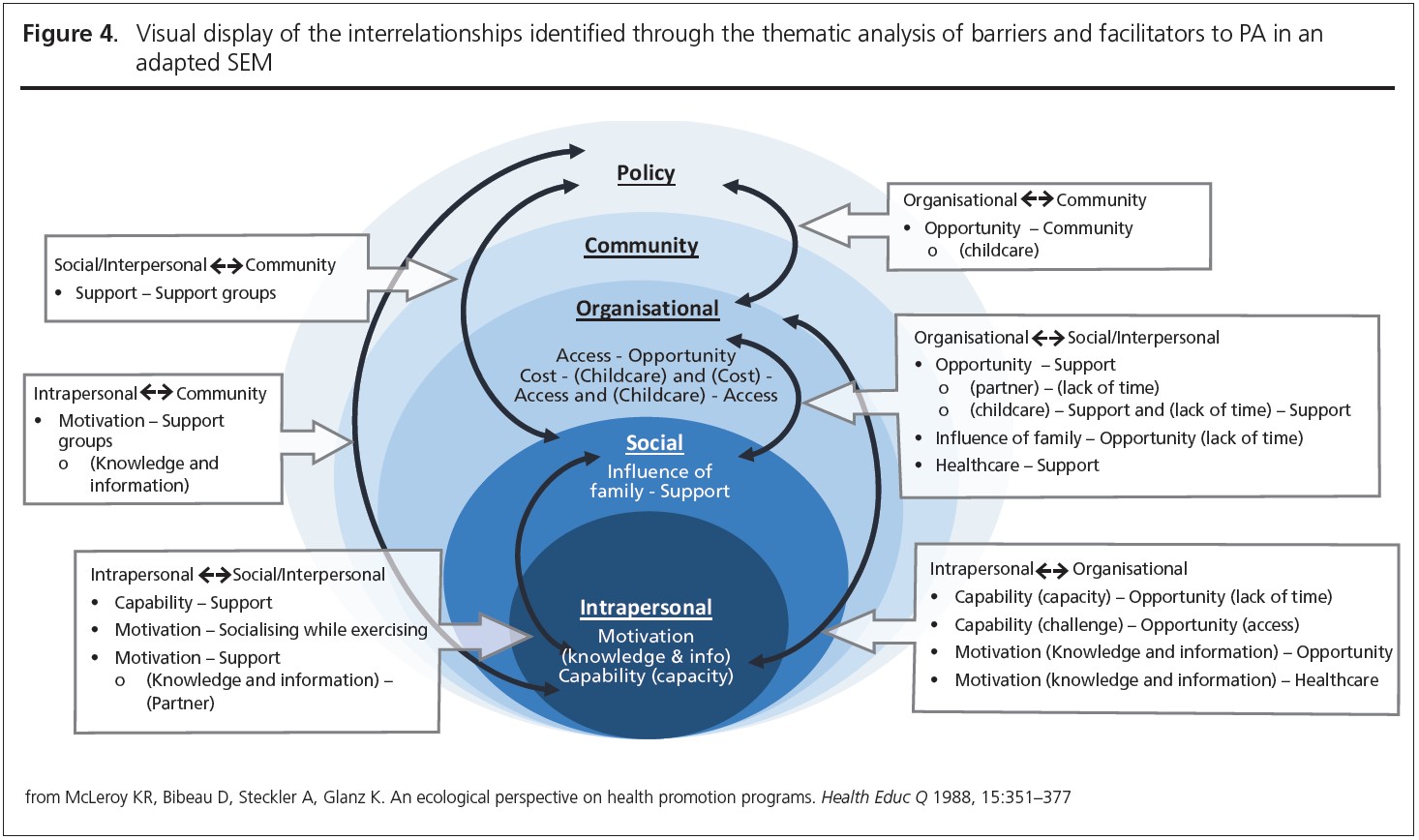
Seven core themes were constructed from the data: two at the intrapersonal (capability and motivation), three at the social (influence of family, socialising while exercising, support) and four at the organisational level (access, opportunity, healthcare, type of exercise). Table 3 provides an overview of papers contributing to each theme. A summary of main themes and example quotes are displayed in Figures 2 and 3.

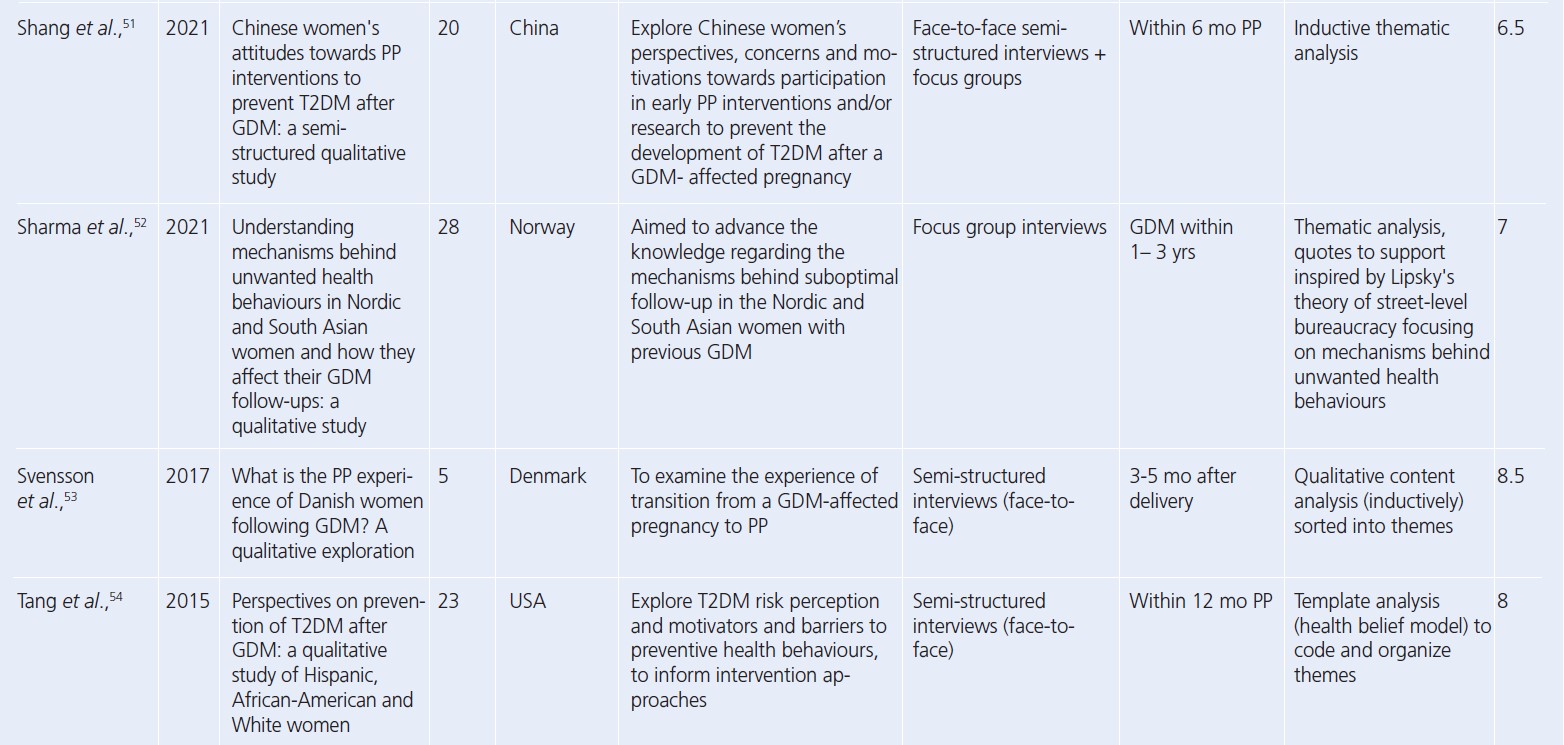

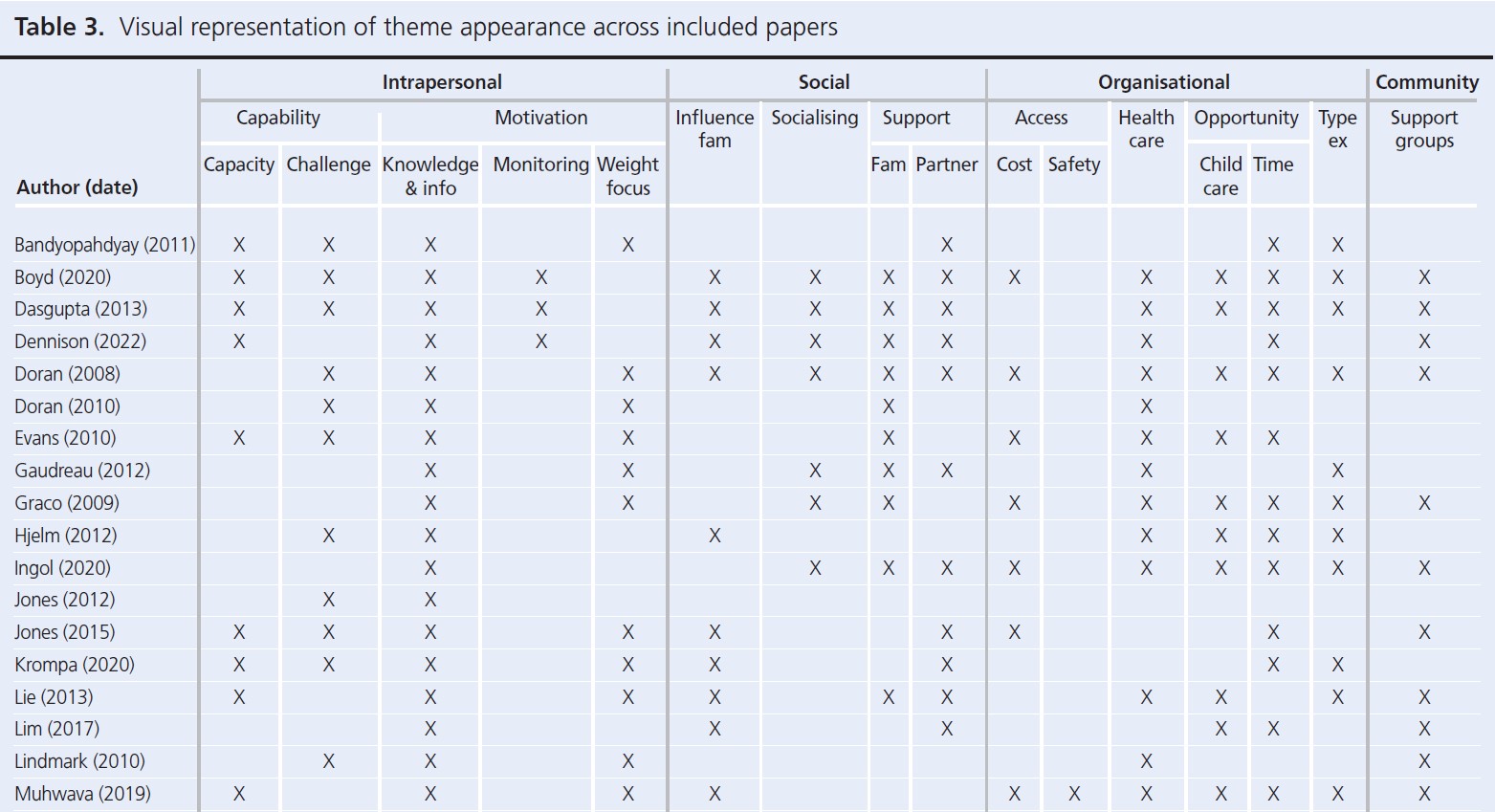
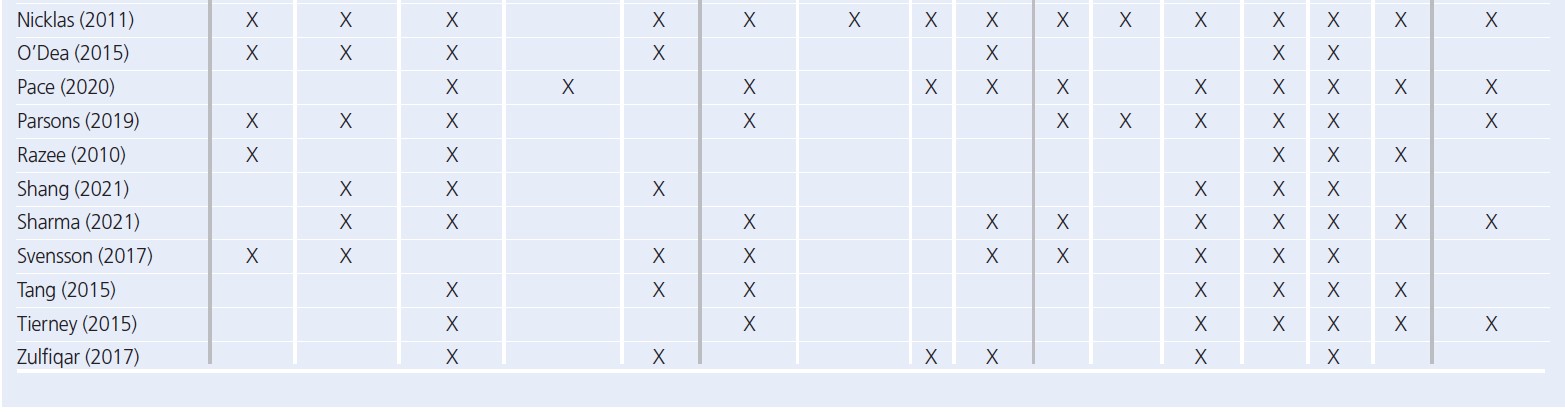
Intrapersonal
Capability referred to whether women felt able to engage with PA. Where PA felt achievable and women were confident, this was facilitative. Motivation related to the desire to engage with PA. While most women were aware of the benefits, they highlighted that this was not sufficient to overcome other barriers. One sub-theme to emerge under motivation was a weight focus, which in the short term was motivating but was debilitative for longer-term, sustainable engagement with PA.
Social
Influence of family was wide-ranging and referred to any effects the family had on PA. For example, commitments and having children were barriers, while role modelling and being well enough to look after children facilitated PA. The presence of support from families, friends and partners was facilitative of PA, while lack of support was a barrier. Partner support was highlighted as vital for engagement with PA. Taking part in PA with other people was also a facilitator to activity.
Organisational
Availability of opportunities, either local resources or provision of activities, facilitated PA while barriers included cost of activities, safety, lack of childcare and other competing demands on time. Leisure PA was the focus, despite the emphasis of a general lack of time or inability to undertake leisure PA. However, participation in activities of daily living, such as domestic (chores, housework) or active travel (walking for transport), were acknowledged as easier to undertake and were prioritised.
Interrelationships between themes
Participant quotes and reported results demonstrated links between every level of the SEM. These interrelationships, highlighted through links between themes and sub-themes, are summarised in Figure 4.
Discussion
To the authors’ knowledge, this is the first review of its kind to classify barriers and facilitators to PA for women with previous GDM according to the SEM. Barriers and facilitators to PA appeared on four levels of the SEM, in addition to interactions within and between these levels, resulting in a complex web of factors that need to be addressed, in combination, for increased PA engagement.
The focus of barriers in the present review was around leisure time purposeful exercise; active transport was identified as achievable and routine.29 The American Diabetes Association recommends that for populations at high risk of T2DM, at least 150 minutes per week of PA should be undertaken.57 Active travel could be one such domain of PA encouraged for women after GDM and is still linked to T2DM prevention.58 Other domains of PA therefore need to be further explored and encouraged, as they may be a more realistic type of PA for women after GDM.
Barriers and facilitators at the social and organisational levels were linked with the interpersonal level, highlighting that behaviour may be compromised by wider barriers.15 Encouraging individual motivation is not sufficient in the presence of higher-level barriers, therefore targeting system-wide approaches, rather than solely individuals, could be more effective.59 It is important to consider these wider factors and the subsequent impact on women’s ability to undertake PA when planning future PA interventions after GDM. Findings in young adult women by Peng et al., including accessibility to PA, familial commitments and the physical environment, overlapped with some of the findings in the present review.16 Defining women by their GDM diagnosis when women feel abandoned postpartum may not be helpful.30 Further research is needed to explore how women after GDM define themselves postnatally, and how they may best be targeted or addressed in the context of PA.
At the individual level, themes capability and motivation align well with the COM-B model of behaviour change, which states that individual behaviour change is influenced by opportunity, motivation and capability.60,61 Within motivation, positioning PA as a method of weight loss was helpful in the short term, but was discouraging for maintaining PA in the long term.45 Women with GDM, and general T2DM prevention advice, are recommended to manage their weight,62,63 which could be debilitative for women with unrealistic expectations for their body and weight postpartum.64 Managing expectations and creating a long-term facilitative PA environment, emphasising broader benefits of PA besides weight loss, could aid longer-term PA uptake and maintenance. Future interventions should therefore still consider individual tailoring and behaviour change theory, in conjunction with addressing wider barriers to PA.

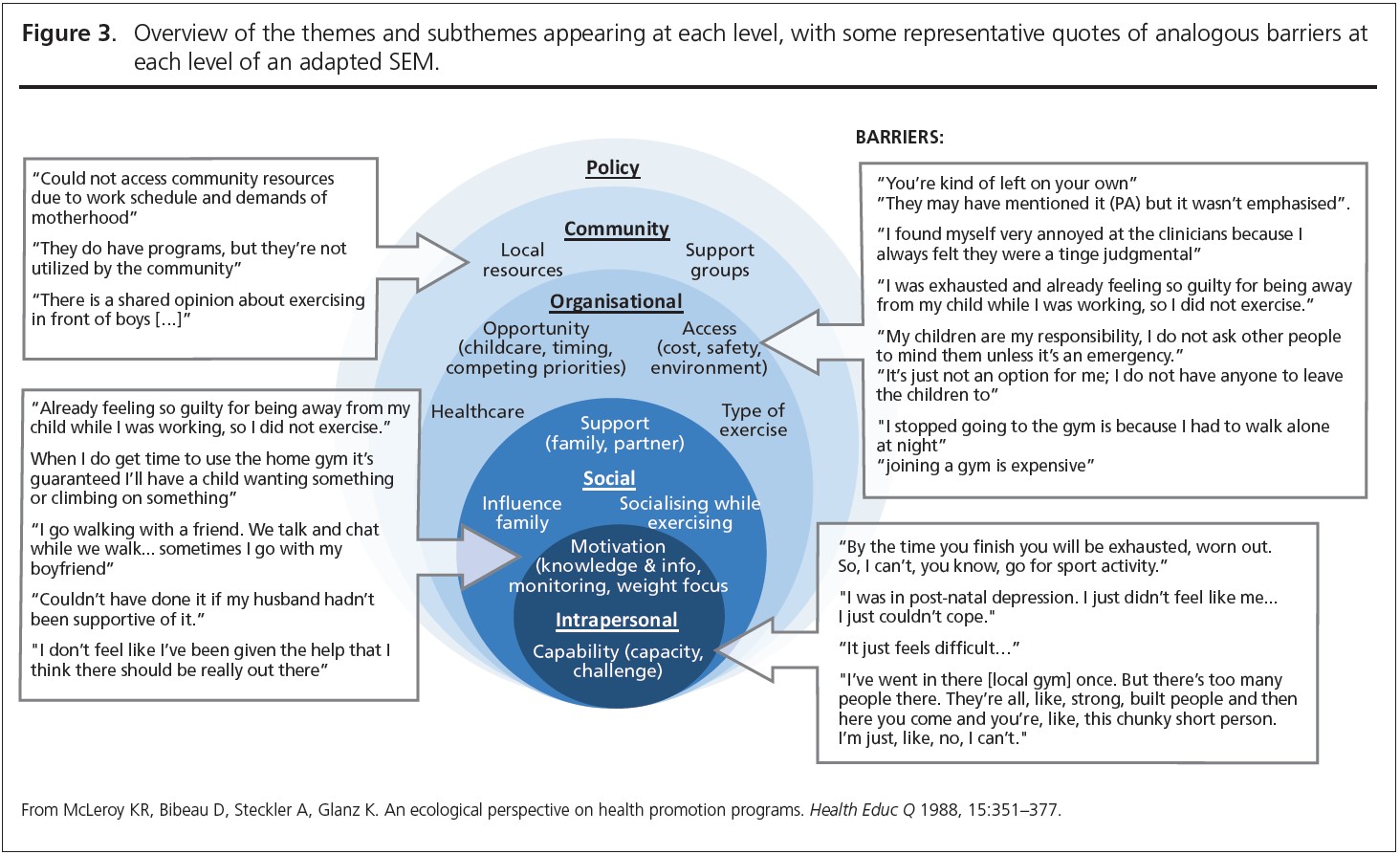
Family commitments are a unique and specific barrier to women after GDM compared to the general population at risk of T2DM. Family-based interventions can increase PA in children,65 which is important since children of women with GDM are at an increased risk of several metabolic disorders, including insulin resistance, T2DM, hypertension and obesity.66 PA can help reduce risk of these metabolic disorders in both mothers and their children.67 Therefore, family-based PA could have multiple benefits across generations. Family-based interventions may also overcome lack of childcare, which was the main organisational barrier identified in the present review. The lack of child-care is a widely cited and known barrier to PA, not just for women after GDM but also for postpartum women in general.68 Further research is needed to establish how childcare could best be provided for maximum uptake and helpfulness to enable engaging with PA. The present review identified that childcare was heavily interrelated with the social level of the SEM. For example, childcare as a barrier was overcome with help from family or partner support,40 and was not overcome when partners were busy or when women did not feel comfortable leaving their children with family for the sake of PA.45,47 Without partner buy-in, PA uptake and maintenance may not be possible for women after GDM. Therefore, PA interventions should consider targeting couples, including partners to ‘tag team’, in addition to other forms of childcare, to increase accessibility of PA for mothers. This is important, as interventions which have addressed childcare when trying to help women be active after GDM could be more successful at increasing PA.69 Providing childcare opportunities in PA contexts is important, not only for women after GDM but at a wider, systems level for all (postnatal) women.
At the social level, support was one of the most quoted factors, posing a barrier when not present but a facilitator when present. Partner support and fostering positive PA environments for the whole family was highlighted as instrumental.31,47,70 When women did not feel supported, they were unable to engage with PA, even if they wanted to. When women did feel supported, or when they had help from their partner or family, they reported more engagement with PA. Partner support specifically, in agreement with Peng et al. was essential in enabling PA.16 Support was also linked with the concept of non-physical community support.45 Creating social ‘community’ and increasing access to PA within communities has been recommended for PA promotion.71 Community-based interventions could be cost-effective methods to increase PA,72,73 including for women with previous GDM.69 Therefore, creating a supportive setting after GDM could partly be achieved by connecting women post- partum. Further research is needed to establish how and what community-based PA intervention could look like, and how it may be implemented for women after GDM.

Strengths and limitations
The SEM helped frame barriers and facilitators according to wider systems, providing more direction for designing multilevel interventions. To the authors’ knowledge, it is also the first review of its kind to consider PA after GDM on a wider systems level. However, the contexts of included studies varied. Extracted results and conclusions could be specific to these contexts, or not generalisable. Additionally, the results synthesised can only shed light on the topic, and it is important that context-specific Patient and Public Involvement (PPI) and/or co-production be included when tailoring or developing interventions.
Conclusion
Women after GDM consistently face wider-level barriers that are not within their direct control to overcome. Reducing the onus on individual mothers by, for example, addressing organisational level barriers like childcare provision, may be important for long- term PA uptake and maintenance. Supplementing individually targeted interventions with wider multi-level population targets should be the focus for future interventions aiming to increase PA in women after GDM.
Conflict of interest None.
Funding This research was funded as part of a Graduate Teaching Assistant Scholarship provided by Sheffield Hallam University. The funder did not have any influence on or direct involvement in the research.
Acknowledgements Sheffield Hallam Librarians aided in the development of search terms and demonstration of database searches for the papers included in the present review.
Data availability Data sharing is not applicable to this article as no datasets were generated or analysed during the current study. All data used were obtained from published articles.
Other information This work was registered on Open Science Framework (https://doi.org/10.17605/OSF.IO/PRG56). For the purpose of open access, the author has applied a Creative Commons Attribution (CC BY) licence to any Author Accepted Manuscript version arising from this submission.
References
- IDF. United Kingdom diabetes report 2000 — 2045 [Internet]. 10th edition IDF Diabetes Atlas, Brussels, Belgium, 2021. Available from: https://diabetesatlas.org/data/en/country/209/gb.html
- Metzger BE. International association of diabetes and pregnancy study groups recommendations on the diagnosis and classification of hyper glycemia in pregnancy. Diabetes Care 2010;33:676–82. Available from: https://pubmed.ncbi.nlm.nih.gov/20190296/ and https://doi.org/10.2337/dc09-1848
- Vounzoulaki E, Khunti K, Abner SC, Tan BK, Davies MJ, Gillies CL. Progression to type 2 diabetes in women with a known history of gestational diabetes: systematic review and meta-analysis. BMJ 2020;369:m1361. https://dx.doi.org/10.1136/bmj.m1361
- Ayman G, Strachan JA, McLennan N, et al. The top ten research priorities in diabetes and pregnancy according to women, support networks and healthcare professionals. Diabet Med 2021;38(8):e14588. https://doi.org/10.1111/dme.14588
- Knowler WC, Barrett-Connor E, Fowler S, et al. Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N Engl J Med 2002;346(6):393–403. https://doi.org/10.1056/NEJMoa012512
- Tuomilehto J, Lindström J, Eriksson JG, et al. Prevention of type 2 diabetes mellitus by changes in lifestyle among subjects with impaired glucose tolerance. N Engl J Med 2001;344(18):1343–50. https://pubmed.ncbi.nlm.nih.gov/11333990/ and https://doi.org.10.1056.NEJM200105033441801
- Pan XR, Li GW, Hu YH, et al. Effects of diet and exercise in preventing NIDDM in people with impaired glucose tolerance: The Da Qing IGT and diabetes study. Diabetes Care 1997;20(4):537–44. https://pubmed.ncbi.nlm.nih.gov/9096977/ and https://doi.org/1-2337/diacare.20.4.537
- NICE. Diabetes in pregnancy overview - NICE Pathways, 2020. Available from: https://pathways.nice.org.uk/pathways/diabetes-in-pregnancy/diabetes-in-pregnancy-overview#content=view-node%3Anodes-postnatal- care-for-women-who-were-diagnosed-with-gestational-diabetes
- Lim S, Chen M, Makama M, O’Reilly S. Preventing type 2 diabetes in women with previous gestational diabetes: reviewing the implementation gaps for health behavior change programs. Semin Reprod Med 2021;1–7. https://pubmed.ncbi.nlm.nih.gov/33511581/ and https://doi.org/10.1055/s-0040-1722315
- Dennison RA, Ward RJ, Griffin SJ, Usher-Smith JA. Women’s views on lifestyle changes to reduce the risk of developing type 2 diabetes after gestational diabetes: a systematic review, qualitative synthesis and recommendations for practice. Vol. 36, Diabetic Medicine 2019;36:702–17. https://doi.org/10.1111.dme.13926
- NHS. NHS England » NHS Diabetes Prevention Programme (NHS DPP), 2019. https://www.england.nhs.uk/ltphimenu/diabetes-prevention/nhs-diabetes-prevention-programme-nhs-dpp/
- Dasgupta K, Terkildsen Maindal H, Kragelund Nielsen K, O’Reilly S. Achieving penetration and participation in diabetes after pregnancy prevention inter- ventions following gestational diabetes: a health promotion challenge. Diabetes Res Clin Pract 2018;145:200–13. http://search.ebscohost.com/login.aspx?direct=true&db=cmedm&AN=29684615&site=ehost-live and https://doi.org/10.1016/j.diabres.2018.04.016
- Stokols D. Translating social ecological theory into guidelines for community health promotion. American Journal Health Promotion 1996;10:282–98. https://pubmed.ncbi.nlm.nih.gov/10159709/ and https://doi.org/10.4278/ 0890-1171-10.4.282
- McLeroy KR, Bibeau D, Steckler A, Glanz K. An ecological perspective on health promotion programs. Health Educ Q 1988;15(4):351–77. https://pubmed.ncbi.nlm.nih.gov/3068205/ and https://doi.org/10.1177/ 109019818801500401
- McGlashan J, Hayward J, Brown A, et al. Comparing complex perspectives on obesity drivers: action‐driven communities and evidence‐oriented experts. Obes Sci Pract 2018;4(6):575–81. https://onlinelibrary.wiley.com/doi/ abs/10.1002/osp4.306 and https://doi.org/10.1002/osp4.306
- Peng B, Ng JYY, Ha AS. Barriers and facilitators to physical activity for young adult women: a systematic review and thematic synthesis of qualitative literature. Int J Behav Nutr Phys Act 2023;20(1):23. https://ijbnpa.biomed- central.com/articles/10.1186/s12966-023-01411-7 and https://doi.org/10.1186/ s12966-023-01411-7
- Jones EJ, Fraley HE, Mazzawi J. Appreciating recent motherhood and culture: a systematic review of multimodal postpartum lifestyle interventions to reduce diabetes risk in women with prior gestational diabetes. Matern Child Health J 2017;21(1):45–57. https://doi.org/10.1007/s10995-016-2092-z
- Gilinsky AS, Dale H, Robinson C, Hughes AR, McInnes R, Lavallee D. Efficacy of physical activity interventions in post-natal populations: systematic review, meta-analysis and content coding of behaviour change techniques. Health Psychol Rev 2015;9(2):244–63. https://doi.org/10.1080/17437199.2014.899059
- Buelo AK, Kirk A, Lindsay RS, Jepson RG. Exploring the effectiveness of physical activity interventions in women with previous gestational diabetes: a systematic review of quantitative and qualitative studies. Prev Med Reports 2019;14:100877. https://linkinghub.elsevier.com/retrieve/pii/ S2211335519300579 and https://doi.org/10.1016/j.pmedr.2019.100877
- Page MJ, McKenzie JE, Bossuyt PM, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ 2021;372:n71. https://www.bmj.com/content/372/bmj.n71 and https://doi.org/10.1136/bmj.n71
- Dennison RA, Fox RA, Ward RJ, Griffin SJ, Usher-Smith JA. Women’s views on screening for type 2 diabetes after gestational diabetes: a systematic review, qualitative synthesis and recommendations for increasing uptake. Diabet Med 2020;37(1):29–43. http://search.ebscohost.com/login.aspx?di-rect=true&db=cmedm&AN=31317569&site=ehost-live and https://doi.org/ 10.1111/dme.14081
- Peacock AS, Bogossian F, McIntyre HD, Wilkinson S. A review of interventions to prevent type 2 diabetes after gestational diabetes. Women and Birth 2014;27:e7–15. https://doi.org/10.1016/j.wombi.2014.09.002
- Goveia P, Cañon-Montañez W, Santos D de P, et al. Lifestyle intervention for the prevention of diabetes in women with previous gestational diabetes mellitus: a systematic review and meta-analysis. Front Endocrinol (Lausanne) 2018;9:583. http://search.ebscohost.com/login.aspx?direct=true&db=cmedm&AN=30344509&site=ehost-live and https://doi.oreg/10.3389/fendo. 2018.00583
- Hewage SS, Wu S, Neelakantan N, Yoong J. Systematic review of effectiveness and cost-effectiveness of lifestyle interventions to improve clinical diabetes outcome measures in women with a history of GDM. Clinical Nutrition ESPEN 2020;35:20–9. http://clinicalnutritionespen.com/article/S240545771930484X/fulltext and https://doi.org/10.1016/j.clnesp.2019.10.011
- Cooke A, Smith D, Booth A. Beyond PICO: the SPIDER tool for qualitative evidence synthesis. Qual Health Res 2012;22(10):1435–43. https://doi.org/10.1177/1049732312452938
- CASP. Critical Appraisal Skills Programme Qualitative Checklist 2018. https://casp-uk.net/images/checklist/documents/CASP-Qualitative-Studies-Checklist/CASP-Qualitative-Checklist-2018_fillable_form.pdf
- Braun V, Clarke V. Reflecting on reflexive thematic analysis. Qualitative Research in Sport, Exercise and Health 2019;11:589-97. https://doi.org/10.1080/2159676X.2019.1628806 and https://www.tandfonline.com/doi/abs/10.1080/2159676X.2019.1628806
- Bandyopadhyay M, Small R, Davey MA, Oats JJN, Forster DA, Aylward A. Lived experience of gestational diabetes mellitus among immigrant South Asian women in Australia. Aust New Zeal J Obstet Gynaecol 2011; 51(4):360–4. https://doi.org/10.1111/j.1479-828X.2011.01322.x
- Boyd J, McMillan B, Easton K, Delaney B, Mitchell C. Utility of the COM-B model in identifying facilitators and barriers to maintaining a healthy post- natal lifestyle following a diagnosis of gestational diabetes: a qualitative study. BMJ Open 2020;10(8):e037318. https://bmjopen.bmj.com/content/10/8/e037318 and https://doi.org/10.1136/bmj.open-2020-037318
- Dasgupta K, Da Costa D, Pillay S, et al. Strategies to optimize participation in diabetes prevention programs following gestational diabetes: a focus group study. PLoS One 2013;8(7):e67878. http://search.ebscohost.com/login.aspx?direct=true&db=cmedm&AN=23861824&site=ehost-live and https://doi.org/10.1371/journal.pone.0067878
- Dennison RA, Griffin SJ, Usher-Smith JA, Fox RA, Aiken CE, Meek CL. “Post- GDM support would be really good for mothers”: a qualitative interview study exploring how to support a healthy diet and physical activity after gestational diabetes. PLoS One 2022;17(1):e0262852 https://doi.org/10.1371/journal.pone.0262852
- Doran F. Gestational diabetes mellitus: perspectives on lifestyle changes during pregnancy and post-partum, physical activity and the prevention of future type 2 diabetes. Aust J Prim Health 2008;13(3):85–92. https://doi.org/10.1071/PY08040
- Doran F, Davis K. Gestational diabetes mellitus in Tonga: insights from healthcare professionals and women who experienced gestational diabetes mellitus. N Z Med J 2010;123(1326):59–67. http://search.ebscohost.com/login.aspx?direct=true&db=cmedm&AN=21326400&site=ehost-live
- Evans MK, Patrick LJ, Wellington CM. Health behaviours of postpartum women with a history of gestational diabetes. Can J Diabetes 2010; 34(3):227–32. https://doi.org/10.1016/S1499-2671(10)43011-7
- Gaudreau S, Michaud C. Cultural factors related to the maintenance of health behaviours in Algonquin women with a history of gestational diabetes. Chronic Dis Inj Can 2012;32(3):140–8. http://search.ebscohost.com/login.aspx?direct=true&db=cmedm&AN=22762900&site=ehost-live
- Graco M, Garrard J, Jasper AE. Participation in physical activity: perceptions of women with a previous history of gestational diabetes mellitus. Health Promot J Austr 2009;20(1):20–5. https://pubmed.ncbi.nlm.nih.gov/19402811/
- Hjelm K, Bard K, Apelqvist J. Gestational diabetes: prospective interview- study of the developing beliefs about health, illness and health care in mi- grant women. J Clin Nurs 2012;21:3244–56. http://search.ebscohost.com/login.aspx?direct=true&db=cmedm&AN=23083394&site=ehost-live and https://doi.org/10.1111/j.1365-2702.2012.04069.x
- Ingol TT, Kue J, Conrey EJ, Oza-Frank R, Weber MB, Bower JK. Perceived barriers to type 2 diabetes prevention for low-income women with a history of gestational diabetes: a qualitative secondary data analysis. Diabetes Educ 2020;46(3):271–8. https://doi.org/10.1177/014572170920255
- Jones EJ, Appel SJ, Eaves YD, Moneyham L, Oster RA, Ovalle F. Cardiometabolic risk, knowledge, risk perception, and self-efficacy among American Indian women with previous gestational diabetes. J Obstet Gy- necol Neonatal Nurs 2012;41(2):246–57. http://search.ebscohost.com/login.aspx?direct=true&db=cmedm&AN=22834848&site=ehost-live and https://doi.org/10.1111/j.1552-6909.2012.01339.x
- Jones EJ, Peercy M, Cedric Woods J, et al. Identifying postpartum intervention approaches to reduce cardiometabolic risk among American Indian women with prior gestational diabetes, Oklahoma, 2012-2013. Prev Chronic Dis 2015;12(4):E45. https://pubmed.ncbi.nlm.nih.gov/25837258/ and https://doi.org/10.5888/pcd12.140566
- Krompa K, Sebbah S, Baudry C, Cosson E, Bihan H. Postpartum lifestyle modifications for women with gestational diabetes: a qualitative study. Eur J Obstet Gynecol Reprod Biol 2020;252:105–11. https://doi.org/10.1016/j.ejogrb.2020.04.060
- Lie MLS, Hayes L, Lewis-Barned NJ, May C, White M, Bell R. Preventing type 2 diabetes after gestational diabetes: Women’s experiences and implications for diabetes prevention interventions. Diabet Med 2013;30(8):986–93. https://pubmed.ncbi.nlm.nih.gov/23534548/ and https://doi.org/10.1111/dme.12206
- Lim S, Dunbar JA, Versace VL, et al. Comparing a telephone- and a group- delivered diabetes prevention program: characteristics of engaged and non- engaged postpartum mothers with a history of gestational diabetes. Diabetes Res Clin Pract 2017;126:254–62. https://dx.doi.org/10.1016/j.diabres.2017.02.026
- Lindmark A, Smide B, Leksell J. Perception of healthy lifestyle information in women with gestational diabetes: a pilot study before and after delivery. Eur Diabetes Nurs 2010;7(1):16–20. https://doi.org/10.1002/edn.150
- Muhwava LS, Murphy K, Zarowsky C, Levitt N. Experiences of lifestyle change among women with gestational diabetes mellitus (GDM): a behavioural diagnosis using the COM-B model in a low-income setting. PLoS One 2019;14(11):e0225431. http://search.ebscohost.com/login.aspx?direct=true&db=cmedm&AN=31765431&site=ehost-live and https://doi.org/10.1371/journal.pone.0225431
- Nicklas JM, Zera CA, Seely EW, Abdul-Rahim ZS, Rudloff ND, Levkoff SE. Identifying postpartum intervention approaches to prevent type 2 diabetes in women with a history of gestational diabetes. BMC Pregnancy Childbirth 2011;11(1):1–8. https://doi.org/10.1186/1471-2393-11-23
- O’Dea A, Tierney M, McGuire BE, et al. Can the onset of type 2 diabetes be delayed by a group-based lifestyle intervention in women with prediabetes following gestational diabetes mellitus (GDM)? Findings from a randomized control mixed methods trial. J Diabetes Res 2015;2015:798460. PMID:26347894 and https://doi.org/10.1155/2015/798460
- Pace R, Loon O, Chan D, et al. Preventing diabetes after pregnancy with gestational diabetes in a Cree community: an inductive thematic analysis. BMJ Open Diabetes Res Care 2020;8(1):e001286. http://search.ebscohost.com/login.aspx?direct=true&db=cmedm&AN=32393481&site=ehost-live and https://doi.org/10.1136/bmjdrc-2020-001286
- Parsons J, Sparrow K, Ismail K, Hunt K, Rogers H, Forbes A. A qualitative study exploring women’s health behaviours after a pregnancy with gestational diabetes to inform the development of a diabetes prevention strategy. Diabet Med 2019;36(2):203–13. http://doi.wiley.com/10.1111/dme.13794 and https://doi.org/10.1111/dme.13794
- Razee H, van der Ploeg HP, Blignault I, et al. Beliefs, barriers, social support, and environmental influences related to diabetes risk behaviours among women with a history of gestational diabetes. Health Promot J Austr 2010;21(2):130–7. http://search.ebscohost.com/login.aspx?direct=true&db=cmedm&AN=20701563&site=ehost-live and https://doi.org/10.1071/he10130
- Shang J, Henry A, Zhang P, et al. Chinese women’s attitudes towards post- partum interventions to prevent type 2 diabetes after gestational diabetes: a semi-structured qualitative study. Reprod Health 2021;18(1):133. https://search.ebscohost.com/login.aspx?direct=true&db=cmedm&AN=34174913&site=ehost-live and https://doi.org/10.1186/s12978-021-01180-1
- Sharma A, Birkeland KI, Nermoen I, et al. Understanding mechanisms behind unwanted health behaviours in Nordic and South Asian women and how they affect their gestational diabetes follow-ups: A qualitative study. Diabet Med 2021;38:e14651. https://onlinelibrary.wiley.com/doi/full/10.1111/dme.14651 and https://doi.org/10.1111/dme.14651
- Svensson L, Nielsen KK, Maindal HT. What is the postpartum experience of Danish women following gestational diabetes? A qualitative exploration. Scand J Caring Sci 2018;32(2):756–64. http://search.ebscohost.com/login.aspx?direct=true&db=cmedm&AN=28856697&site=ehost-live and https://doi.org/10.1111/scs.12506
- Tang JW, Foster KE, Pumarino J, Ackermann RT, Peaceman AM, Cameron KA. Perspectives on prevention of type 2 diabetes after gestational diabetes: a qualitative study of Hispanic, African-American and White women. Matern Child Health J 2015;19(7):1526–34. https://link-springer-com.hallam.idm.oclc.org/article/10.1007/s10995-014-1657-y and https://doi.org/10.1007/s10995-014-1657-y
- Tierney M, O’Dea A, Danyliv A, et al. Factors influencing lifestyle behaviours during and after a gestational diabetes mellitus pregnancy. Health Psychol Behav Med 2015;3(1):204–16. https://doi.org/10.1080/21642850.2015.1073111
- Zulfiqar T, Lithander FE, Banwell C, et al. Barriers to a healthy lifestyle post gestational-diabetes: an Australian qualitative study. Women Birth 2017; 30(4):319–24. https://doi.org/10.1016/j.wombi.2016.12.003
- Colberg SR, Sigal RJ, Yardley JE, et al. Physical activity/exercise and diabetes: a position statement of the American Diabetes Association. Diabetes Care 2016;39:2065-79. https://diabetesjournals.org/care/article/39/11/2065/37249/Physical-Activity-Exercise-and-Diabetes-A-Position and https://doi.org/10.2337/dc16-1728
- Saunders LE, Green JM, Petticrew MP, Steinbach R, Roberts H. What are the health benefits of active travel? A systematic review of trials and cohort studies. PloS One 2013;8(8):e69912. https://doi.org/10.1371/journal.pone.0069912
- Rutter H, Savona N, Glonti K, et al. The need for a complex systems model of evidence for public health. Lancet 2017;390:2602–6. https://doi.org/10.1016.S0140-6736(17)31267-9
- Michie S, van Stralen MM, West R. The behaviour change wheel: a new method for characterising and designing behaviour change interventions. Implement Sci 2011;6(1):1–42. https://implementationscience.biomedcentral.com/articles/10.1186/1748-5908-6-42 and https://doi.org/10.1186/1748-5908-6-42
- West R, Michie S. A brief introduction to the COM-B Model of behaviour and the PRIME theory of motivation. Qeios 2020 Apr 9. https://doi.org/10.32388/WW04E6.2
- NICE. Type 2 diabetes: prevention in people at high risk; 1.13 Weight management advice. NICE guidelines, 2017. https://www.nice.org.uk/guidance/PH38/chapter/Recommendations#weight-management-advice
- NICE. Diabetes in pregnancy: management from preconception to the postnatal period. NICE, 2020. https://www.nice.org.uk/guidance/ng3/chapter/Recommendations#postnatal-care
- Hodgkinson EL, Smith DM, Wittkowski A. Women’s experiences of their pregnancy and postpartum body image: a systematic review and meta- synthesis. BMC Pregnancy Childbirth 2014;14(1):330. https://doi.org/10.1186/1471-2393-14-330
- Brown HE, Atkin AJ, Panter J, Wong G, Chinapaw MJM, van Sluijs EMF. Family-based interventions to increase physical activity in children: a systematic review, meta-analysis and realist synthesis. Obes Rev 2016;17(4): 345–60. https://pubmed.ncbi.nlm.nih.gov/26756281/ and https://doi.org/10.1111/obr.12362
- Bianco ME, Josefson JL. Hyperglycemia during pregnancy and long-term off- spring outcomes. Curr Diab Rep 2019;19(12):143. PMCID:PMC7008468 https://doi.org/10.1007/s11892-019-1267-6
- Rush E, Simmons D. Physical activity in children: prevention of obesity and type 2 diabetes. Med Sport Sci 2014;60:113–21. https://pubmed.ncbi.nlm.nih.gov/25226806/ and https://doi.org/10.1159/000357341
- Makama M, Awoke MA, Skouteris H, Moran LJ, Lim S. Barriers and facilitators to a healthy lifestyle in postpartum women: A systematic review of qualitative and quantitative studies in postpartum women and healthcare providers. Obes Rev 2021;22(4):e13167. https://search.ebscohost.com/login.aspx?direct=true&db=cmedm&AN=33403746&site=ehost-live and https://doi.org/10.1111/obr.13167
- Ioannou E, Humphreys H, Homer C, Purvis A. A systematic review using the socio-ecological model for physical activity interventions aiming to prevent type 2 diabetes after gestational diabetes (abstract only). Journal Sports Sciences;2022:7. https://www.tandfonline.com/doi/abs/10.1080/02640414.2022.2125766
- Barnett I, Guell C, Ogilvie D. How do couples influence each others' physical activity behaviours in retirement? An exploratory qualitative study. BMC Public Health 2013;13(1):1197. https://doi.org/10.1186/1471-2458-13-1197
- Heath GW, Parra DC, Sarmiento O, et al. Evidence-based intervention in physical activity: lessons from around the world. Lancet 2012;380;272–81. PMCID:PMC4978123. https://doi.org/10.1016/S0140-6736(12)60816-2
- Roux L, Pratt M, Tengs TO, et al. Cost effectiveness of community-based physical activity interventions. Am J Prev Med 2008;35(6):578–88. https://doi.org/10.1016/j.amepre.2008.06.040
- Bock C, Jarczok MN, Litaker D. Community-based efforts to promote physical activity: a systematic review of interventions considering mode of delivery, study quality and population subgroups. J Sci Med Sport 2014;17(3):276–82. https://doi.org/10.1016/j.jsams.2013.04.009